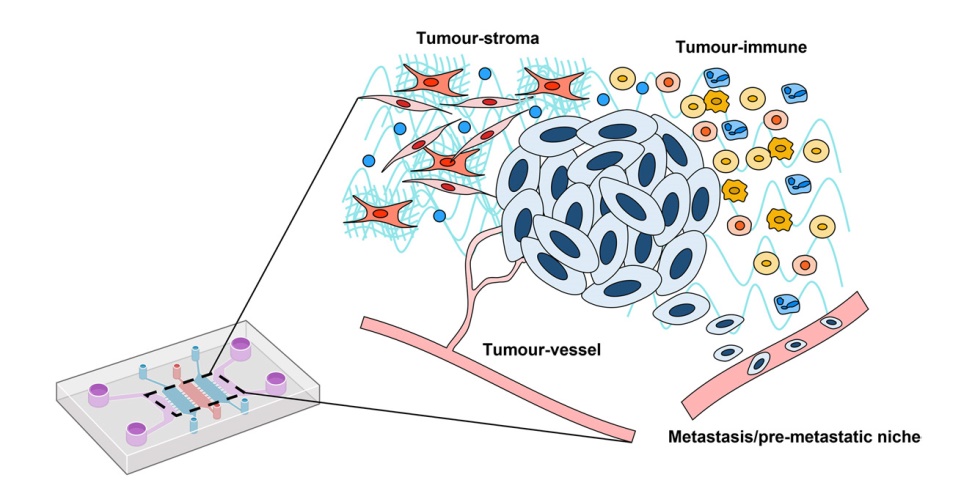
The pro-inflammatory, immunosuppressive microenvironment is a hallmark of the TME. However, more insight on the effects of the TME on host immune cells is needed. Ayuso et al. developed a 3D in vitro tumour-on-a-chip platform to examine how NK cells respond to an immunosuppressive microenvironment [
112]. The tumour-on-a-chip device consisted of a central microchamber where MCF-7 breast cancer cells were embedded in collagen with or without NK cells [
112]. The asymmetric distribution of nutrients can generate multiple tumour scenarios and created gradients of cell viability (
Fig. 4B(i.)). As a result of the heterogenous distribution of nutrient and oxygen, immune cells isolated from the chip expressed genes associated with immune exhaustion (indoleamine 2,3-dioxygenase (IDO1), programmed cell death protein 1 (PD-1) and cytotoxic T-lymphocyte-associated protein 4 (CTLA-4)), as well as nutrient starvation and hypoxia (HIF-1A, VEGF) were upregulated (
Fig. 4B(i.)) [
112]. Genes associated with cell survival, proliferation and activation (B-cell lymphoma 2 (BCL2), granzyme B (GZMB), Fas cell surface death ligand (FASL), IL-15 were significantly downregulated, suggesting an immunosuppressive tumour-on-a-chip microenvironment (
Fig. 4B(ii.)) [
112]. On the other hand, the NK cells isolated from the well-plate had a gene expression profile similar to naive NK cells (NK cells that have never been exposed to cancer cells), compared to the profile NK cells from the microdevice [
112]. More specifically, NK cells cultured in well plates exhibited a significant upregulation of activation/ prosurvival markers, whereas NK cells from the microdevice exhibited an upregulation of genes associated with NK cell exhaustion [
112]. Treatment with IDO1 and PD-1 inhibitors led to partial alleviation of immune exhaustion and an increase in MCF-7 cell death [
112]. This platform allowed for the study of molecular alteration that drives immunosuppression and to identify new immunotherapeutic targets. Additionally, it highlights the importance of analyzing molecular changes of immune cells in a physiologically relevant platform, as well as the spatial dependence on cancer cell response to immunotherapies, as tumour cells in the necrotic region were observed to be highly resistant to treatment. Aung et al. developed a perfusable tumour-ona- chip model to study the effects of breast cancer-immune interactions on T-cell recruitment [
115]. Breast cancer cells, monocytes and endothelial cells were spatially controlled in a gelatin matrix using 3D micropatterning [
115]. The effects of cancer-monocyte interactions on T-cell recruitment were examined by dispersing T-cells into the perfused culture medium and allowed to infiltrate through the endothelial barrier [
115]. Microfluidic culture simulating the hypoxic environment commonly found in solid tumours/spheroids resulted in increased T-cell recruitment, compared to dispersed cancer cells [
115]. Addition of monocytes into the system also resulted in higher T-cell recruitment, attributed to differences in chemokine secretion profiles that influence endothelial permeability [
115]. Overall, this platform allowed for the study of immune cell recruitment using multiple cell types.