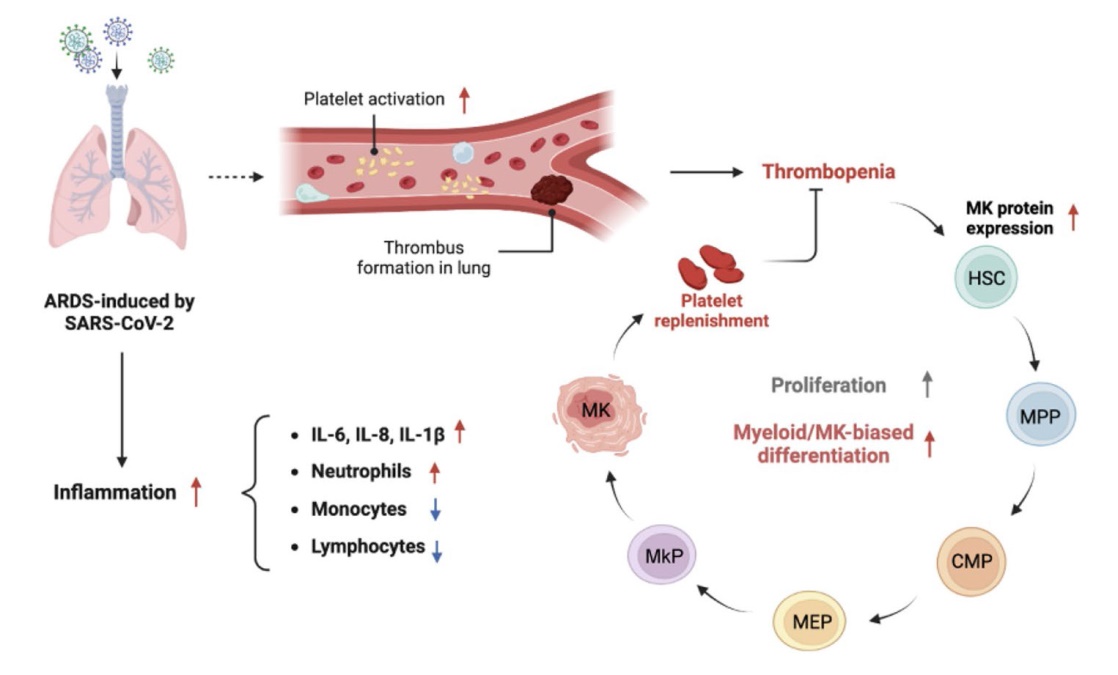
To interpret the internal changes in HSC/MPP compartment under COVID-19-induced ARDS, we sorted Lin− CD34+CD38−CD45RA− cells and employed RNAsequencing to evaluate the transcriptome response. RNA-seq identified 395 differentially expressed genes (DEGs), 18 of which are highly related to platelet function (
Fig. 4A). Notably, platelet adhesion-related gene
MMP8 and activation-related gene
OLR1 were significantly elevated in ICU-ARDS samples which further proved the existence of increased thrombus burden. Besides,
LCN2, a gene expressed in platelet and could promote the proliferation of CD34
+ cells, was highly expressed in ICUARDS samples, demonstrating that early in HSC/MPP cells there is a tendency of active platelet regeneration. However, genes representing platelet-biased or MK-biased HSCs were downregulated in ICU-ARDs samples, including
CLU,
SDPR, and
CD9, suggesting a highly active state of platelet regeneration from these HSC populations under the COVID-19 ARDS inflammation state to replenish the consumed platelets. Consistent with this, the proportion of CD41
+ HSCs/MPPs was reduced in the systemic inflammation state (Supplementary Fig. 2A), while the average CD41 expression level on the surface of CD41-positive HSCs/MPPs was significantly increased (Supplementary Fig. 2B), indicating higher MK protein synthesis. Heatmap analysis revealed that all ICU-ARDS groups were significantly clustered and distinguished from the non- COVID-19 group (
Fig. 4B). Besides, we sorted out the pathways related to hematopoiesis and inflammation in the gene ontology (GO) database and summarized the corresponding gene sets to further look for potential associations. There were 50 hematopoietic-cell-lineage-related genes and 60 inflammatory-response-related genes overlapped with DEGs and 26 DEGs were highly related to both hematopoiesis and inflammation, illustrating the connection between systemic inflammation and hematopoietic disorder under ARDS induced by severe COVID-19 infection (
Fig. 4C). Furthermore, GO analysis illustrated that several platelet- and myeloid-related pathways were enriched in ICU-ARDS groups, demonstrating the trend of myelopoiesis and platelet production in the HSC/MPP compartment (
Fig. 4D). Interestingly, HSC/MPP cells affected by severe COVID-19 infection exhibited more sensitivity to platelet-derived growth factors (GO:0036119 and GO0036120). Given that platelets in ICU-ARDS patients were in a ready-to-activate state and platelet activation directly led to the release of platelet derived growth factor (PDGF), PDGF produced by hyperactivated platelet may contribute to the elevated proliferation ability of HSC/MPP cells in ARDS patients. Furthermore, gene-set enrichment analysis (GSEA) based on GO gene sets revealed stem-cell-development- and immune-related pathways were significantly enriched, further indicating the abnormal activity of HSCs/MPPs in ICU-ARDS patients (
Fig. 4E-G). However, antigen processing and presentation and type I interferon signaling pathway were inhibited in ICU-ARDS samples, demonstrating the immunosuppression under ARDS induced by COVID-19 which was in accordance with the systemic T cell reduction (
Fig. 3B). In summary, RNA-seq analysis illustrated that HSC/MPP cells in ICU-ARDS patients exhibited elevated proliferation ability and incline of megakaryocytic/platelet lineage differentiation as a response to ARDS caused by severe COVID-19 infection.