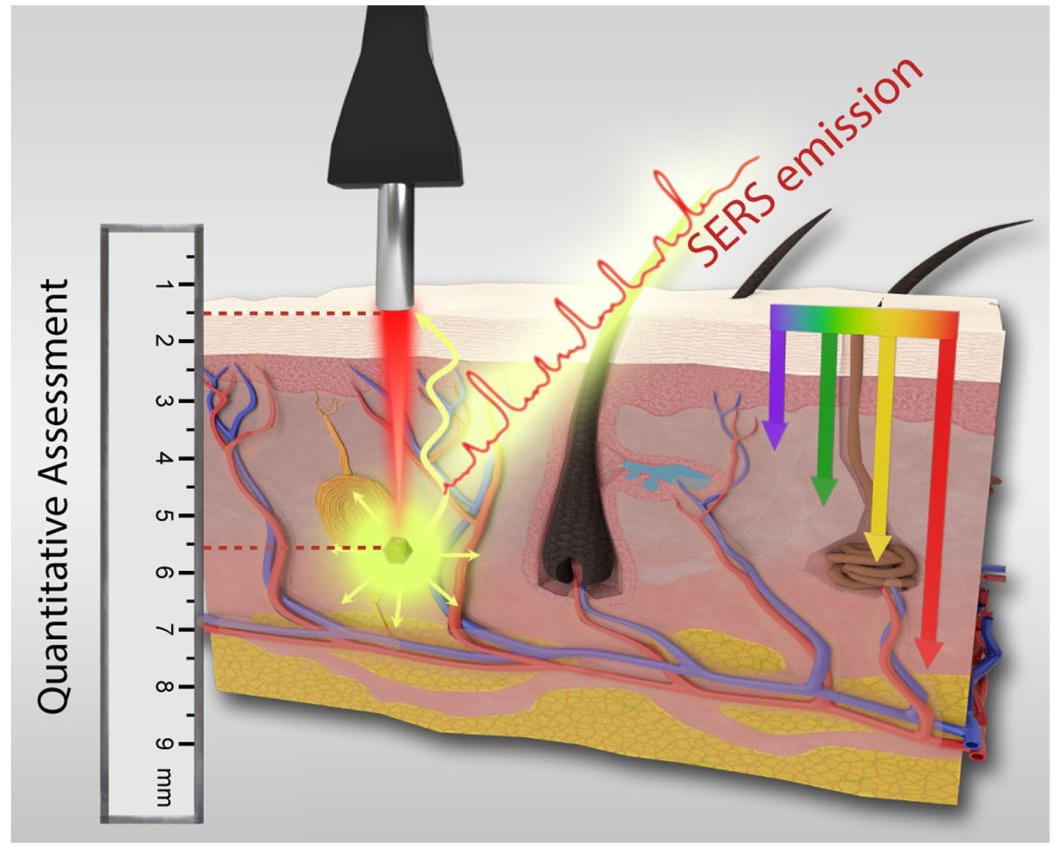
Before our detection depth assessments, we conducted the optimization of the laser beam focal plane. With the light-tissue interactions, a shift of laser focal plane may occur.
Figure 2e shows the effects of light-tissue interactions [
25]. First is reflection at the interface (
Fig. 2e, i), such as the tumor-normal tissue interface for the in vivo environment; then comes refraction when light enters a tissue structure with a different refractive index (
Fig. 2e, ii); there is also intense absorption of photon energy (
Fig. 2e, iii) and multiple scattering of photons (
Fig. 2e, iv); the autofluorescence is usually caused by the biological chromophores (
Fig. 2e, v). Refraction and scattering are believed to induce the shift of laser focal plane, with the former bends light, and the latter randomizes the photon propagation direction. To determine that how these interactions would affect the focal plane and spectral intensity, our optimization procedure consists of the sequential acquisitions of Raman spectra on tissue-covered SERS gels while tuning the distance from the laser probe to gel surface. The focal depth and focal length of the laser beam was measured as 6.8 and 1.22 mm, respectively (Fig. S6). As a pure SERS gel was put under the laser beam, the strongest signal was obtained when the probegel distance is 4.9 mm (Fig. S7). This indicated that the optimal focus position of the pure SERS gel was ~ 1.9 mm under the gel surface. This is reasonable, considering the common cases that the best laser focus is usually beneath the material surface. We then placed porcine muscle tissues with a thickness of 1- 6 mm on the gel. As shown in
Fig. 2f, with 1 mm of muscle tissue on top, the optimal probe-gel distance extends; the optimal focal plane is in the range of 5.91-6.89 mm, with the highest SNR obtained at 6.7 mm, very close to the focal length of 6.8 mm (
Fig. 2f). Here, the optimal focal plane could be regarded as the distorted focal length of the laser beam, while it does not follow the Gaussian distribution due to the tissue light diffusion. With 2 or 3 mm muscle tissues on top, the curves become flatter, and the optimal focal planes further extend to 6.61-7.0 mm and 6.91-7.3 mm, respectively. We have also conducted the same measurements on fat and skin tissues (Fig. S8), and similar phenomenon were observed, i.e., the flattening of the SNR distribution profile as well as the tendency of the shift in optimal focal plane. As mentioned above, this could be explained by the interface refraction or tissue scattering. Refraction leads to an extended focal length, since the refractive index of tissues (
n =
1.4) is higher than that of the air (
n =
1); Multiple scattering of photons results in light diffuse and defocus (
Fig. 2g). For thick tissues, scattering plays a more important role and can be described by the physical parameter, transport mean free path (TMFP). TMFP is the mean propagation distance that it takes for photons to lose relation to the propagation direction they had before entering the tissue. It takes into account the average angle by which photons are scattered in each scattering event. Depending on the tissue type and wavelength of operation, TMFP in muscle at the NIR region (650-1000 nm) was reported to be ~ 1.1 mm [
26]. Therefore, for thick tissues of ≥ 2 mm, the incidence no longer follows the shape of a Gaussian beam, and the SNR becomes less sensitive to the probe-gel distances (as shown in
Fig. 2f). When the tissue thickness increases to 3—6 mm, it is hardly able to tell the optimal focal plane. All the measurements were repeatedly conducted at least 3 times on different batches of tissues and SERS gels. The above results demonstrated that the optimal probe-gel distances are close to the focal length. To ensure the consistency of measurements, in the following studies, we fixed the distances between probe and gel surface the same as the focal length.