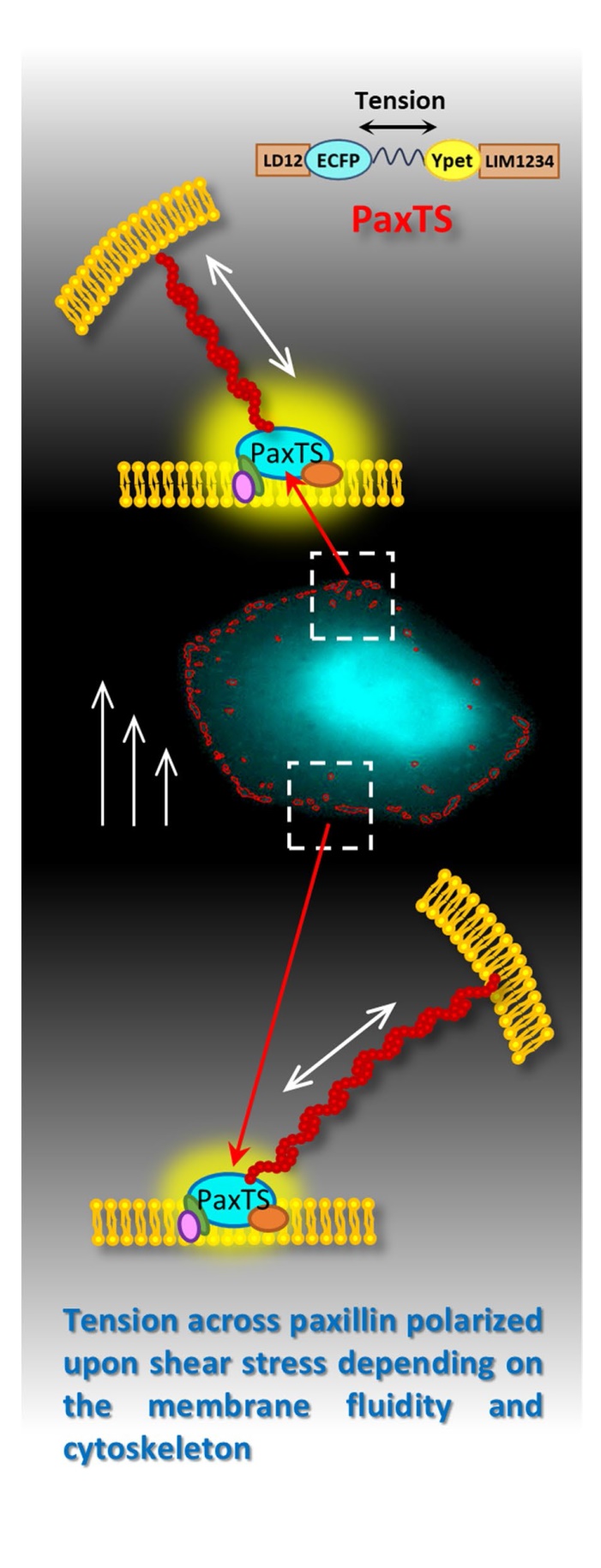
Different components of cytoskeleton are tightly linked and align along the direction of shear stress, which is the most significant mechanosensitive step in intracellular force transmission [
32]. As one component of cytoskeleton, actin filaments are anchored to lipid rafts as well as running across the cell and terminating at FAs [
33]. The LD domains of paxillin link it to actin filaments by combining with actopaxin and other actin-binding proteins [
34,
35]. As assumed above, the force that can be transmitted to paxillin by cytoskeleton is the core in the shear stress-induced polarized distribution of tension across paxillin. Undoubtedly, the force can transmit along cytoskeleton [
12,
36,
37], and the connection between paxillin and actin filaments provides a possibility for direct force transmission. To access this, drugs were applied to cells, and our results showed that destroying the structure of the cytoskeleton abolishes the tension difference between upstream and downstream regions. It revealed that cytoskeleton indeed participates in the regulation of tension polarity across paxillin upon shear stress, and the function of microfilaments and microtubules are similar, although paxillin is linked to actin filaments but indirectly to microtubules. Interestingly, our results also indicated that inhibiting the contractility of actomyosin suppressed the tension polarity across paxilin upon shear stress. However, when considering the role of microfilaments, the dynamic of actin filaments should not be ignored. The contractility of actomypsoin acts as buffers to resist the force applied on cells. Inhibiting the myosin decreasing the cell stiffness [
38]. Soft cells deform more significantly and asymmetrically between the upstream and downstream regions upon shear stress, applying a larger force on the upstream regions. The actin network without contractility is insufficient to resit the force. Hence, the tension endured by paxillin is different between the upstream and downstream regions, once the contractility of actomyosin was inhibited. In general, these results conform to our assumption that shear stress-induced polarity of tension across paxillin is related to cytoskeleton. Furthermore, both the integrity and the contractility of cytoskeleton play important roles in the process.