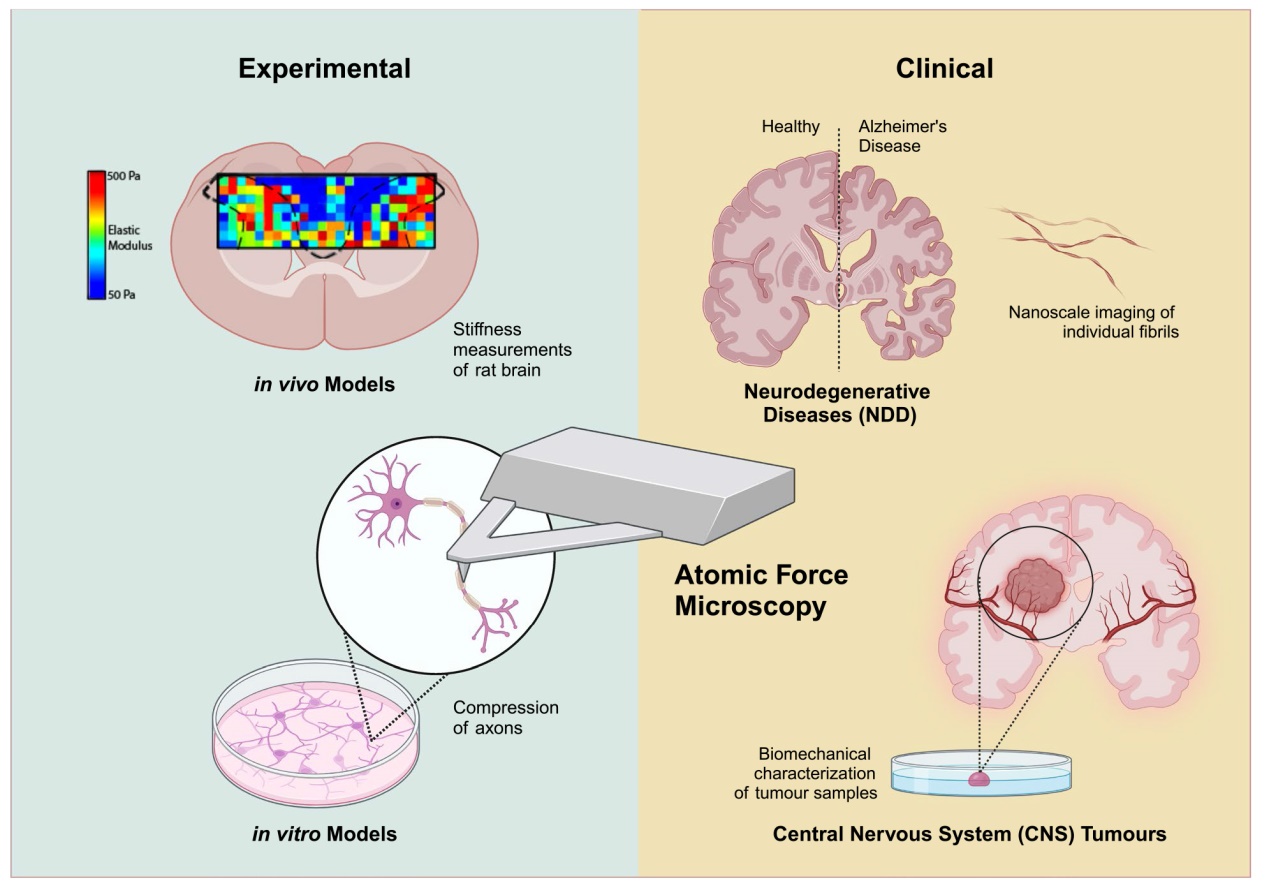
Introduction
AFM characterization techniques
AFM operation
Fig. 1 a AFM operation principles. AFM utilizes a sharp tip at the free end of a soft cantilever to scan across a sample surface. Height measurements at discrete points create a reconstruction of the sample's topography. b, c Various AFM-based techniques used for characterizing biological samples. Schematics of the force-distance (FD) curves are shown. b The probe indents the sample until it reaches a defined maximum force (blue approach curve) before retracting (black retraction curve) back to its rest position. Stiffness values can be obtained by fitting the slope of the approach curve (a steeper slope corresponds to higher stiffness). The hysteresis or sample viscosity can be estimated from the area between the approach and retraction curves. c The adhesion force (red dot) can be extracted from the retraction curve. Single receptor-ligand interactions can be measured when the AFM tip is functionalized with bioligands |
Morphology
Elastic properties
Viscoelastic properties
Molecular interaction
AFM characterization of neurological disorders using experimental models
Fig. 2 Schematics illustrating AFM-based BK probing [32] a Schematic showing the functionalization of the AFM tip with a BK antibody. b Schematics showing the functionalized probe detecting BK channels and FD curves obtained in the presence and absence of BK channels. CM denotes cell membrane. c Representative force-distance curves observed under the various conditions. d Microscopy image and schematic showing the use of an AFM to detect BK channels on neurons using a functionalized probe. e Representative AFM heatmaps displaying specific BK probe binding events over 1 μm2 areas. Colours represent the measured force of specific binding events. f Graphs comparing the unbinding force and BK channel density between WT and KO neurons |
Fig. 3 Axonal compression experimental model [20]. a (i) A bead attached to the tip of the AFM cantilever was used to gradually compress (sub-nN force) the DRG and hippocampal axons cultured in parallel channels. The axonal response to compression was influenced by the duration and magnitude of the applied force. After releasing the compression force, the four responses include : (ii) axons recovered to the original state, (iii) axons remained permanently deformed, (iv) axons entered a degenerative process characterized by increased focal axonal swelling (FAS), and (v) in severe cases, axonal rupture occurred. DRG axons are more resistant to compression than hippocampal axons. b After compression with 540 Pa for 10 min, DRG axons completely recovered. However, at higher pressures, FAS was resulted along the whole axon (arrows). c Single hippocampal axons were compressed with pressures ranging from 65 - 550 Pa for 10 - 30 mins. Axonal shape and mitochondria transport were not recovered after compression with pressures > 65 Pa for 10 mins. Each panel represents one axon at different time points, before (*), during, and after compression (**). Lower panels denotes 3x zoom of the compression region of the axons before and after compression release. Scale bar : 10 μm |
Fig. 4 Neurons grow toward soft tissue [22]. a Schematic depicting how local gradients in brain tissue stiffness perpendicular to the RGC axon growth direction (M) and the local optic tract (OT) curvature (C) were determined. b Axons turned toward the softer side of the tissue preferentially in vivo. Data plot showing relationship between M and C. c Same data as (B) represented in a bar chat. n : number of measurements from 7 animals. d-e Time-lapse imaging of individual axon bundles growing on a stiffness gradient that mimicked in vivo conditions revealed that in vitro, in the absence of chemical gradients, RGC axons preferentially turned toward the softer side of the substrate. Scale bar : 20 μm. f Eye primordium cultured on a similar stiffness gradient (colour) showed that axons grow more clockwise in the left half and more counterclockwise in the right half of the image, demonstrating a preference for turning toward the softer side of the substrate. Scale bar : 200 μm. g Quantification of individual axon segment orientations confirmed preferential turning toward the soft side of the substrate |
Emerging clinical applications of AFM in neurological disorders
Neurodegenerative diseases
Alzheimer's disease
Table 2 Notable applications of AFM in the evaluation of clinical samples from NDD patients |
 |
Fig. 5 Evaluation of Clinical Samples of NDD Patients Using AFM (a) Schematics of the collection of CSF and the subsequent deposition of protein fibrils on gold substrate within a liquid cell for AFM imaging. b Phase-contrast AFM image showing three distinct conformation of protein fibrillar aggregates in either parallel (green), sliced (red) or T-junction (blue). c-e High resolution AFM height, phase and overlayed images of individual fibrils detected in the CSF of AD patient. f AFM height image of ultralong single fibrils. Adapted and modified from [45]. g The statistical plot, overlayed with a Lorentzian fit, of the mean height of spherical aggregate (red) and the mean height of fibrillar aggregate (grey) on the Red blood cellf (RBC) surface (right insert). h A schematic showing the protein aggregate assembly on RBCs as a function of increasing patient age and decreasing cognition. Adapted and modified from [16]. i-j Representative AFM images of serum from PD patient and healthy control showing extensive presence of aggregates in PD serum. Adapted and modified from [53]. k-n Representative AFM images of platelets from healthy individual (k) and patients with PD (l), ALS (m) and AD (n). Adapted and modified from [55] |
Parkinson's disease
Amyotrophic lateral sclerosis
Central nervous system tumours
Table 3 Notable clinical applications of AFM in the evaluation of CNS tumour samples |
 |
Challenges and limitations
Table 4 Challenges and limitations of AFM use in clinical applications |
 |